Doppler ultrasound and gray-scale are the first and primary imaging tool for the liver transplantation assessment of the graft,
follow-up and detection of complications.
Ultrasound (US) is cost-effective,
accessible,
noninvasive,
and easily performed at bedside,
allowing an adequate assessment of liver parenchyma,
vascularization and bile duct.
However,
this method has limitations and when US findings are inconclusive,
imaging with other modalities is necessary.
The examination should include US- gray scale of the liver parenchyma and color and pulsed Doppler of the hepatic artery,
portal vein,
suprahepatic veins and inferior vein cava (IVC).
Multidetector CT an MRI allows the evaluation of parenchymal and vascular structures and they are accurate and noninvasive methods for evaluating the hepatic vessels.
Cross-sectional modalities,
such as CT and MR imaging,
are commonly used to confirm suspicions aroused by Doppler US findings or to exclude a clinical hypothesis when US results are normal or inconclusive.
The most common and most significant postoperative complications after liver transplantation are vascular complications (arterial and venous stenoses and thromboses) and/or non-vascular (biliary strictures,
leakage,
abscesses,
bilomas and posttransplant lymphoproliferative disease).
- VASCULAR COMPLICATIONS: They usually occur in the early postoperative period and rapid diagnosis is critical for graft survival.
A resistive index (RI) less than 0.5 is associated with vascular complications.
A hepatic artery (HA) velocity of 50–200 cm/s and an RI of 0.5–0.8 are associated with normal graft status in children within the first year after transplant.
Vascular and biliary complications are higher with HA velocity range of 201–300 cm/s.
In order to maximize the diagnosis of vascular complications it is important to recognize anatomical vascular variants in both the donor and their recipient,
as well as the surgical anastomotic techniques utilized.
HEPATIC ARTERY: HA–related complications are the most common liver transplant–related vascular complications (12–25% of pediatric recipients and account for 60% of posttransplant vascular complications).
A significant reduction in arterial supply may result in parenchymal ischemia and infarctions,
bile leak,
formation of intrahepatic biloma,
and possibly liver abscesses that may lead to graft failure.
The RI of a normal hepatic artery is 0.5– 0.8.
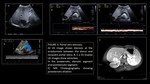
- HEPATIC ARTERY STENOSIS: (Figure 2)
It has been reported to occur in 5%–11% of liver transplant recipients and usually
occurs at the site of anastomosis within 3 months after transplantation.
At the site of narrowing,
spectral analysis reveals a focal accelerated velocity greater than 2 m/sec.
However,
the diagnosis is usually made distal to the stenosis.
Intrahepatic arterial waveforms distal to the stenosis display a tardus parvus pattern with a decreased resistive index (< 0,5).
Parvus tardus pattern may be a normal finding during 48- 72 hours after transplantation.
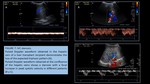
- HEPATIC ARTERY THROMBOSIS: (Figure 3)
Is more common in split or living donor liver transplantation with an incidence around 7%.
Complete occlusion of the hepatic artery results in infarction or necrosis of the liver parenchyma and may lead to fulminant hepatic failure in the early posttransplantation period.
US-based diagnosis of hepatic artery thrombosis is established in the absence of flow in the hepatic and intrahepatic artery at color and pulsed Doppler imaging.
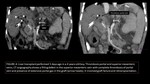
Spiral CT or MRI is also useful in confirming or excluding hepatic artery thrombosis.
The criterion for a diagnosis of hepatic artery thrombosis at both CT and MR angiography is the appearance of an abrupt cut-off of flow in the artery,
usually at the site of the anastomosis.
PORTAL VEIN:
- PORTAL VEIN THROMBOSIS: (Figure 4)
Portal vein thrombosis occurred in about 1-2% of cases.
Risk factors include surgical technique,
hypercoagulable state,
decreased portal venous inflow,
an increase of the hepatic resistance secondary to outflow impediment or excessive vessel redundancy among others.
The US shows echogenic clot and color flow and spectral Doppler analysis will show no detectable flow within the portal vein.
At contrast material–enhanced CT,
portal vein thrombosis is seen as a low-attenuation filling defect.
When the thrombus extends to the periphery of the intrahepatic portal venous branches,
it can no longer be treated with balloon dilation or thrombolysis,
and the patient must undergo another transplantation.
- PORTAL VEIN STENOSIS: (Figure 5 y 6)
Portal vein stenosis is a rare complication,
has a reported incidence of 1% after liver transplantation.
At gray-scale US,
portal vein stenosis is diagnosed when a reduction of the vessel lumen of 50% or more is observed or when the caliber of the vessel is 2.5 mm or less at the site of narrowing.
Color Doppler US shows focal color aliasing at the vascular anastomosis and the waveform shows a peak anastomotic velocity greater than 125 cm/sec or a velocity in the stenotic segment that is three to four times greater than that in the prestenotic segment.
Turbulent flow may be a normal finding in the early postoperative period.
CT angiography may be performed to quantify the degree of stenosis.
HEPATIC VEINS AND IVC STENOSIS OR THROMBOSIS:
Complications of the IVC and the hepatic vein have a low combined incidence (<1%) and may occur in the immediate postoperative course due to thrombosis or technical factors.
The normal Doppler waveform obtained in the hepatic vein in a liver transplant is triphasic because of the effect of pressure variations determined by the right cardiac chambers during the cardiac cycle.
The loss of the usual expected normal triphasic spectral waveform among hepatic veins is an indirect sign of outflow obstruction.
IVC stenosis is more common in the pediatric population and it occurs,
many times,
secondary to an anastomotic size discrepancy.
Gray-scale US demonstrates reduction of the caliber of the IVC at the anastomosis and at the same time,
the pulsed Doppler US shows a three to fourfold increase in velocity through the stenosis relative to that in the prestenotic segment,
associated color Doppler aliasing and absence of phasicity in the IVC.
A persistent monophasic waveform is a sensitive finding,
but it is not specific for a substantial hepatic vein stenosis.
(Figure 7)
IVC stenosis and hepatic vein stenosis may manifest as Budd-Chiari syndrome,
with hepatomegaly,
ascites,
reversed flow or absence of phasicity in the hepatic veins,
and reversed flow in the portal vein.
Thrombosis of the hepatic vein or IVC may be depicted as an intraluminal echogenic thrombus with no flow on Doppler images.
CT Coronal imaging and MR are useful for determining the extent of IVC thrombosis.
- BILIARY COMPLICATIONS (Figure 8,
9 y 10)
Biliary complications are the more frequent complications following pediatric liver transplantation and they may occur approximately 25% of patients,
usually with in the first 3 months.
Most of the biliary complications,
before the stage of biliary necrosis,
could be reversible with timely reinstitution of the blood supply.
Ischemic arterial events may result in bile duct strictures or leaks,
increasing the risk of cholangitis,
sepsis,
and abscess.
Advanced biliary ischemia and necrosis are incompatible with graft survival,
consequently a retransplant is required.
Complications include biliary leaks and stricture,
intrahepatic bile duct stones or sludge and biloma.
MR cholangiography is the best noninvasive technique for evaluation of the biliary tree.
Anastomotic strictures often require surgical or radiologic intervention.
Percutaneous transhepatic cholangiography can also be performed for initial treatment,
including balloon dilation,
drainage,
and stent placement.
US shows dilated intrahepatic ducts with dilatation of the proximal bile duct to the level of the anastomosis.
Bile leaks are associated with high morbidity and mortality and the approximate incidence is 5%.
Fluid collections are usually well recognized at US.
MR cholangiography,
performed with specific contrast agents,
can be used in the diagnosis of small bile leak.
A small bile leak may close spontaneously but occasionally surgery is necessary.
Formation of sludge or stones may be related with alterations to bile associated with stasis and biliary strictures,
retained stones within the graft or cyclosporine treatment.
When the transplanted hepatic artery blood supply is insufficient,
bile duct necrosis develops,
possibly leading to hepatic parenchymal infarction,
bilomas,
or abscesses (Figure 11).
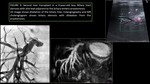
Infarcts may manifest in the US as geographic lesions,
which are often solid and contain central hypoechoic areas that represent liquefaction and necrosis.
At CT we can observe irregular and wedge-shaped low-attenuation lesions located primarily in the periphery of the liver.
The lesions are either unenhanced or heterogeneously enhanced.
Intrahepatic bilomas (Figure 12) usually manifest at US or CT as intrahepatic peripheral cystic lesions that communicate with the bile duct.
Differentiation between biloma and abscess is often difficult.
Fluid collections and hematomas are common in the areas of vascular and biliary anastomosis.
The usually are found during the first days after transplantation and disappear within a few weeks.
Attention should be paid to any increase in size or change in echogenicity of these collections,
findings that may indicate bleeding or infection.
Neoplasms: Risk factors for the development of malignancies in liver transplants include long-term immunosuppression,
previous viral infection (with a hepatitis virus,
Epstein-Barr virus,
cytomegalovirus,
or herpesvirus),
and acute rejection episodes.